Ventilator-associated Pneumonia - StatPearls - NCBI …
35 hours ago · Ventilator-associated pneumonia is a lung infection that develops in a person who is on a ventilator. A ventilator is a machine that is used to help a patient breathe by giving oxygen through a tube placed in a patient’s mouth or nose, or through a hole in the front of the neck. An infection may occur if germs enter through the tube and get ... >> Go To The Portal
Are We wrong when we make the initial diagnostic diagnosis of VAP?
Likewise, all of the early diagnostic indicators of VAP have poor test performance (e.g., fever, leukocytosis, sputum production, and chest radiograph). Consequently, we are doomed to usually be wrong when we make the initial diagnosis of VAP.
Why is VAP associated with critically ill patients?
Critically ill patients, patients with catheters or monitoring devices, vented patients, and patients in the ICU. 41. Why is VAP associated with intubated patients? Because they are at a high risk of aspiration, the cuff may not fully protect the airway, folds present in the cuff allow for secretions from the mouth to enter the lung.
How is ventilator-associated pneumonia (VAP) diagnosed?
Diagnosis of ventilator-associated pneumonia can be difficult. It requires a combination of clinical, radiographic, and microbiologic data. The longer the length of intubation, the higher the likelihood of ventilator-associated pneumonia.
What four radiological changes may be seen in VAP?
What four radiological changes may be seen in VAP? (1) Alveolar shadowing, (2) air bronchograms, (3) fissure abutment, and (4) new chest x-ray infiltrates. 39. What would the role of a biomarker in VAP?
How do you confirm a VAP?
Diagnosing VAP requires a high clinical suspicion combined with bedside examination, radiographic examination, and microbiologic analysis of respiratory secretions. Aggressive surveillance is vital in understanding local factors leading to VAP and the microbiologic milieu of a given unit.
What is VAP and the statistics?
VAP is the second most common nosocomial infection and the leading cause of death from nosocomial infections in critically ill patients 9. Its incidence ranges from 5% to 67% depending on case mix and the diagnostic criteria used 10, and the highest rates are in immunocompromised, surgical, and elderly patients.
What is Hcap diagnosis?
The definition of HCAP includes hospitalization for two days or more within the preceding 90 days, residence in a nursing home or extended care facility, the use of home infusion therapy (including antibiotics), receipt of chronic dialysis within 30 days, home wound care and a history of infection with a multidrug- ...
What is the VAP protocol?
The VAP is defined as a respiratory tract infection developed after 48 hours of intubation with mechanical ventilation or within 48 hours after disconnecting the ventilator.
What is VAP prevention?
To reduce risk for VAP, the following nurse-led evidence-based practices are recommended: reduce exposure to mechanical ventilation, provide excellent oral care and subglottic suctioning, promote early mobility, and advocate for adequate nurse staffing and a healthy work environment.
How do you calculate vent days?
Thus, if 25 patients were ventilated during the month and, for purposes of example, each was on mechanical ventilation for 3 days, the number of ventilator days would be 25 x 3 = 75 ventilator days for February. The Ventilator-Associated Pneumonia Rate per 1,000 Ventilator Days then would be 12/75 x 1,000 = 160.
What is HCAP and HAP?
Healthcare Associated Pneumonia As many of us who have been practicing within the last decade have known, the entity of healthcare-associated pneumonia [HCAP] necessitated similar empiric therapy to that of hospital-acquired pneumonia [HAP] and ventilator-associated pneumonia [VAP].
How is HAP diagnosed?
The IDSA/ATS guidelines recommend non-invasive sputum sampling, such as endotracheal aspirate to diagnose HAP rather than invasive sampling such as bronchoscopy. They also recommend against using procalcitonin, C-reactive protein, and CPIS score for diagnosis. The evidence for blood cultures is controversial.
What is the difference between HAP and VAP?
Hospital-acquired (or nosocomial) pneumonia (HAP) is pneumonia that occurs 48 hours or more after admission and did not appear to be incubating at the time of admission. Ventilator-associated pneumonia (VAP) is a type of HAP that develops more than 48 hours after endotracheal intubation.
What are the nursing interventions for managing the mechanically ventilated patient?
Caring for the Mechanically Ventilated PatientMaintain a patent airway. ... Assess oxygen saturation, bilateral breath sounds for adequate air movement, and respiratory rate per policy.Check vital signs per policy, particularly blood pressure after a ventilator setting is changed.More items...
Why is VAP prevention important?
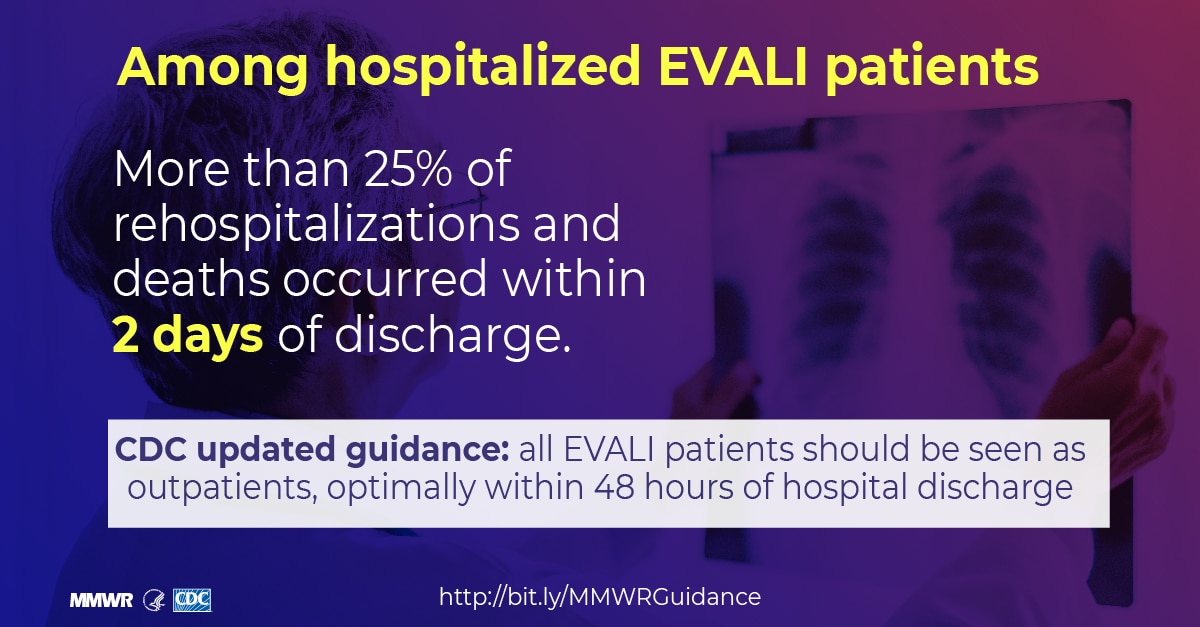
It is the most fatal of the hospital acquired infections, with higher mortality rates than either central line infections or sepsis. Ventilated patients who develop VAP have mortality rates of 45 percent, compared to 28 percent for ventilated patients who do not develop VAP.
What are the nursing responsibilities related to the care of a patient on a ventilator?
Top 10 care essentials for ventilator patientsCare essential 1: Review communications. ... Care essential 2: Check ventilator settings and modes. ... Ventilator settings and modes. ... Care essential 3: Suction appropriately. ... Care essential 4: Assess pain and sedation needs. ... Care essential 5: Prevent infection.More items...•
What is a VAP?
Ventilator-associated Pneumonia (VAP) minus. Related Pages. Ventilator-associated pneumonia is a lung infection that develops in a person who is on a ventilator. A ventilator is a machine that is used to help a patient breathe by giving oxygen through a tube placed in a patient’s mouth or nose, or through a hole in the front of the neck.
What is the CDC's goal for ventilator-associated pneumonia?
CDC provides guidelines and tools to the healthcare community to help end ventilator-associated pneumonia and resources to help the public understand these infections and take measures to safeguard their own health when possible.
How to calculate VAP per 1000 ventilator days?
The VAP rate per 1000 ventilator days is calculated by dividing the number of VAPs by the number of ventilator days and multiplying the result by 1000 (ventilator days).
Where does a pedVAP surveillance occur?
Surveillance may occur in any inpatient pediatric location where denominator data can be collected, such as critical/intensive care units (pedICUs), specialty care areas (SCA), step-down units, wards, and long-term care units. In-plan surveillance for ventilator-associated pneumonia (pedVAP) using the criteria found in this chapter is restricted to patients of any age in pediatric locations (excludes neonatal locations). In-plan surveillance conducted for mechanically-ventilated patients in adult locations (regardless of age) will use the Ventilator-Associated Event (VAE) protocol (see VAE chapter). The PNEU definitions are still available for those units seeking to conduct off-plan PNEU surveillance for mechanically-ventilated adult, pediatric and neonatal patients and non-ventilated adult, pediatric or neonatal patients. The PNEU definitions are also available for secondary bloodstream infection assignment when performing Central Line-Associated Bloodstream Infection (CLABSI) surveillance in ventilated or non-ventilated patients in any location. A complete listing of inpatient locations and instructions for mapping can be
How to calculate ventilator utilization ratio?
The Ventilator Utilization Ratio is calculated by dividing the number of ventilator days by the number of patient days. These calculations will be performed separately for the different types of ICUs, SCAs, and other locations in the institution.
How are device days and patient days used?
Device days and patient days are used for denominators (see Key Terms chapter). Ventilator days, which are the number of patients managed with a ventilatory device, are collected daily, at the same time each day, according to the chosen location using the appropriate form (CDC 57.116, 57.117, and 57.118). These daily counts are summed and only the total for the month is entered into NHSN. Ventilator days and patient days are collected for each of the locations where VAP is monitored. When denominator data are available from electronic sources (for example: ventilator days from respiratory therapy), these sources may be used as long as the counts are not substantially different (+/- 5%) from manually-collected counts, validated for a minimum of three months.
Can you analyze data in NHSN?
All data that is entered into NHSN can be analyzed at event or summary level. The data in NHSN can be visualized and analyzed in various ways, specifically, descriptive analysis reports for both the denominator and numerator data.
What is the diagnosis of VAP?
Often-applied clinical criteria for the diagnosis of VAP are the presence of a new lung infiltrate on chest radiography plus at least two of the following: fever > 38°C, leukocytosis or leukopenia, and purulent secretions.
How many patients with VAP will have VAP?
Ten to 20% of patients who require mechanical ventilation for > 48 h will acquire VAP, with mortality rates of 15 to 50%.
What is a standardized diagnostic algorithm?
A standardized diagnostic algorithm employing clinical and microbiologic data are used in the National Nosocomial Infection Surveillance System (NNIS) to facilitate the application of consistent criteria in reporting nosocomial pneumonia. The clinical pulmonary infection score (CPIS) [which uses microbiologic data] or a modified CPIS (which does not use microbiologic data) have also been proposed to improve diagnostic consistency among clinicians and investigators. A CPIS > 6 is often regarded as consistent with a diagnosis of pneumonia.
What are the sources of clinical information for VAP?
There are roughly four sources of clinical information which should be rapidly available, when considering the possibility of VAP. Try to consider all evidence, rather than anchoring on a single bit of evidence. Additionally, trends in vital signs and data are often more informative than any single data point (since VAP occurs in the context of critical illness, we will always know the patient's baseline values).
How many people are diagnosed with VAP after antibiotics?
Among patients who are started on antibiotics for VAP, only ~40-50% are eventually diagnosed with it after all the data is available.( 18091545, 31754887 ) Therefore, it's essential to re-consider the diagnosis as additional information becomes available. If emerging evidence is inconsistent with a diagnosis of pneumonia, antibiotics should be discontinued and the diagnosis should be discarded.
What is the sensitivity of bronchoscopic specimens?
Bronchoscopy with protected specimen brush and quantitative culture has only a 61% sensitivity and 77% specificity (for a positive likelihood ratio of 2.6 and a negative likelihood ratio of 0.5). ( 32306086 ) Clinicians may overestimate the value of hard-earned bronchoscopic data, leading to misdiagnosis.
What to do if hospital water system is Legionella?
If your hospital water system is known to harbor Legionella, add coverage for it (e.g., azithromycin) and obtain a Legionella urinary antigen test.
How long after intubation does VAP occur?
the riddle of VAP. Ventilator associated pneumonia (VAP) is pneumonia occurring more than two days after intubation. Clinicians must walk several fine lines regarding VAP: Undertreatment: Overlooking the diagnosis until the patient develops septic shock.
How long does it take for infiltrates to disappear?
If infiltrates rapidly disappear (within <24-48 hours), this suggests atelectasis rather than pneumonia. In true pneumonia, infiltrates take many days to weeks to resolve.
Is lung ultrasonography useful for VAP?
Lung ultrasonography. Ultrasonography is less useful for VAP than for community -acquired pneumonia, because many patients in the ICU will have dependent consolidation and B-lines due to atelectasis. However, ultrasonography may remain useful (e.g., to distinguish pleural effusion vs. consolidation at the lung bases).
Why is it so frustrating to diagnose VAP?
That’s because the patients are already intubated and in a serious condition and you really do not want or need to cause any more harm.
How to treat VAP?
Basically, you treat VAP with proper antibiotics. Medications that are prescribed should match the bacteria that is present. Initially, while no information about the causative agent is available, a broad-spectrum antibiotic would be administered until culture and sensitivity results are available.
How to prevent ventilator pneumonia?
About one-third of all ventilator-associated pneumonia cases in the United States could be prevented. For healthcare providers, the CDC released some of the things that we can do to prevent VAP. This includes: 1 Use NPPV (non-invasive positive pressure ventilation) whenever possible, as this significantly decreases the chances of VAP as opposed to intubation and mechanical ventilation. 2 Use daily weaning trials to assess the patient’s respiratory muscle strength and ability to be removed from the ventilator. The risk of VAP is associated with the length of time on the ventilator, so naturally, if you can decrease that amount of time, you will decrease the chances of VAP. 3 Elevate the head position of the bed. This has been shown to reduce the chances of gastric aspiration, which causes VAP. Strive to keep the head of the bed elevated to 45 degrees. 4 Use proper hand hygiene. This goes without saying, however, you should always wash your hand with soap and water upon entering the patient’s room, then again once you leave the room. 5 Practice oral decontamination on the patient by doing mouth-care regularly. Cleaning out the bacteria in the mouth decreases that chances of VAP significantly. 6 Don’t break the circuit of the ventilator. Keeping the circuit closed as much as possible has been shown to decrease the chances of the patient obtaining VAP. This means that you should change the circuit only when it is visibly soiled.
How long does it take for a VAP to develop?
The first one is the early-onset VAP which happens 48 to 96 hours (4 days) after intubation. It is caused by an antibiotic-sensitive bacterium. The second is late-onset VAP, which develops after 4 days from when the patient is initially intubated. It is usually caused by an antibiotic-resistant bacterium.
What does VAP mean in medical terms?
Let’s dig into it a little deeper. VAP stands for Ventilator-Associated Pneumonia and is basically pneumonia that develops 48 hours or longer after mechanical ventilation is initiated on a patient.
How can ventilator-associated pneumonia be acquired?
These pathogens can be acquired through contaminated medical equipment, hands, and even the uniforms of medical providers.
What is VAP in medical terms?
Ventilator-associated pneumonia (VAP) is a term used to describe pneumonia (lung infection) that develops in a patient who has been on mechanical ventilation for more than 48 hours.
What are the symptoms of ventilator-associated pneumonia?
Ventilator-associated pneumoniaincludes clinical signs of purulent tracheal discharge, fevers, respiratory distress, and micro-biological signs of the presence of microorganisms along with white blood cells in the tracheal aspirate along withradiological evidence of pneumonia.
What is ventilator pneumonia?
Ventilator-associated pneumonia is the second most common hospital-acquired infection among pediatrics and neonatal intensive care unit patients. It accounts for 7% to 32% of healthcare-associated infections and 10% of all pediatric device-related infections reported to the National Healthcare Safety Network (NHSN).
What are the symptoms of tracheitisis?
Tracheitisis the presence of clinical signs of purulent tracheal discharge, fevers, respiratory distress, and the presence of bacteria and white blood cells in the tracheal aspirate withoutradiological signs of pneumonia.
Is pneumonia in neonates inversely proportional to birth weight?
In neonates, the rate of ventilator-associated pneumonia is inversely proportional to birth weight.
How to diagnose VAP?
VAP can be accurately diagnosed by any one of several standard criteria: histopathologic examination of lung tissue obtained by open lung biopsy, rapid cavitation of a pulmonary infiltrate in the absence of cancer or tuberculosis, positive pleural fluid culture, same species with same antibiogram isolated from blood and respiratory secretions without another identifiable source of bacteremia, and histopathologic examination of lung tissue at autopsy ( 4 ). However, these criteria are based on invasive procedures for obtaining lung tissue or on uncommon manifestations or complications of VAP. Given the invasive nature of lung biopsy and the infrequent occurrence of other manifestations used as standard criteria, another approach is needed for the definitive diagnosis of VAP. In 1979, a fiberoptic bronchoscopic technique was introduced for obtaining uncontaminated lower respiratory tract secretions, which were cultured quantitatively ( 5 ). The causative microorganisms were recovered at >10 3 CFU/mL from six patients with clinical evidence of lower respiratory tract infection.
What is a VAP patient?
Ventilator-associated pneumonia (VAP) is defined as nosocomial pneumonia in a patient on mechanical ventilatory support (by endotracheal tube or tracheostomy) for >48 hours. For many years, VAP has been diagnosed by the clinical criteria published by Johanson et al. in 1972, which include the appearance of a new or progressive pulmonary infiltrate, fever, leukocytosis, and purulent tracheobronchial secretions ( 1 ); however, these criteria are nonspecific ( 2 ). In the mechanically ventilated patient, fever may be caused by a drug reaction, extrapulmonary infection, blood transfusion, or extrapulmonary inflammation. Pulmonary infiltrates may be due to pulmonary hemorrhage, chemical aspiration, pleural effusion, congestive heart failure, or tumor. Both fever and pulmonary infiltrates occur in the fibroproliferation of late acute respiratory distress syndrome, atelectasis, and pulmonary embolism, as well as in VAP. Cultures of tracheal aspirates are not very useful in establishing the cause of VAP ( 2 ). Although such cultures are highly sensitive, their specificity is low even when they are cultured quantitatively ( 3 ).
Why is VAP more common in ARDS than in other causes of respiratory failure?
VAP is more common in patients with ARDS than in those with other causes of respiratory failure ( 26,32,33 ); it occurs later and is caused by more resistant microorganisms. The diagnosis of VAP is more difficult in such patients because ARDS and VAP have very similar clinical manifestations. Chastre et al. observed no significant differences in temperature, leukocyte count, Pao 2 /Fio 2 ratio, or radiologic score in patients with ARDS with and without VAP ( 32 ). Since clinical criteria for VAP lack both sensitivity and specificity in patients with ARDS, microbiologic data are thought to play a prominent role in the diagnosis of VAP that complicates ARDS ( 26 ). In a study of the use of bronchoscopically directed BAL to diagnose VAP in patients with ARDS, bronchoscopic findings modified antibiotic therapy in 91% of patients with positive BAL cultures and prevented the use of new antibiotics in 54% of patients with insignificant growth ( 33 ). Given the severity of illness of patients with ARDS, particularly when complicated by VAP, and the great difficulty in differentiating VAP from ARDS on clinical and radiographic grounds, the most effective approach to diagnosis of VAP in patients with ARDS is quantitative culture and microscopic examination of lower respiratory tract secretions.
What is the best bronchoscopic technique for VAP?
Two bronchoscopic techniques have been introduced for the accurate diagnosis of VAP in the absence of standard criteria. The protected specimen brush (PSB) collects 0.001 mL of lower respiratory tract secretions and has a diagnostic threshold of >10 3 CFU/mL ( 8 ). BAL, an unprotected technique, samples approximately one million alveoli and has a diagnostic threshold of >10 4 CFU/mL ( 8 ). A protected BAL technique with a balloon-tipped catheter has also been described ( 9 ). Detection of >5% of neutrophils or macrophages with intracellular organisms on a Wright-Giemsa stain of a smear of cytocentrifuged BAL fluid is also diagnostic of VAP ( 10 ).
What is repeatability in bronchoscopy?
Repeatability, which is defined as the variation in repeated measurements of the same quantity ( 28 ), is one measure of the accuracy of a technique in diagnosing the diseases (s) for which it was developed. Marquette and associates performed a study in which a single investigator performed bronchoscopy on 22 patients with suspected VAP ( 28 ). At each bronchoscopy, five successive PSB samples were taken from the same area of the lung. All PSB specimens were cultured quantitatively by the same technologist. In each patient, all five PSB procedures identified exactly the same microorganisms. In 59% of the patients, there was more than a 1-log variation in quantitative culture of the five PSB specimens; in 3 (13.6%) of the 22 patients, quantitative culture results were spread out on both sides of the 10 3 CFU/mL breakpoint. Thus, in spite of the substantial variability of the quantitative cultures, all five PSB procedures for 19 (86.4%) of 22 patients gave results on the same side of the breakpoint, indicating acceptable repeatability.
What is the correlation between pneumonia and bronchoalveolar lavage fluid?
In 1987, a correlation was observed between pneumonia and >10 5 CFU/mL in bronchoalveolar lavage (BAL) fluid ( 6,7 ). Kahn and Jones noted that BAL fluid with >10 5 CFU/mL and <1% squamous epithelial cells had 100% sensitivity and specificity for the diagnosis of bacterial pneumonia.
What is the best test for VAP?
In conclusion, in the absence of gold standard criteria for the diagnosis of VAP, the diagnostic test of choice is quantitative culture and microscopic examination of lower respiratory tract secretions. This approach provides the most accurate diagnosis of VAP and identification of the causative microorganism (s), can predict the onset of VAP and provide the identity and susceptibility of the causative microorganism (s) at the time clinical manifestations of VAP appear, can be used to assess the cause of therapy failure, provides the most effective modality for diagnosis of VAP that complicates ARDS, minimizes misclassification of cases of VAP for studies on the epidemiology of VAP, and minimizes the selective pressure for development of resistant microorganisms. Whether this approach to the diagnosis of VAP has an effect on outcome and reduces deaths is yet to be determined.